
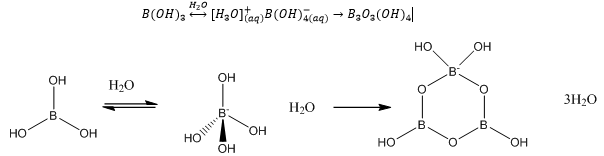

22 mei 2018 — DO NO. T WRITE IN THIS AREA. (d) (i) Ammonia and boron trifluoride react to form a compound NH3BF3 which contains a dative covalent bond.
The only possibility for boron is to bond to three hydrogen atoms, in which case it forms a compound. (borane, BH3) that does not fulfill the octet rule.. This is unusual for hydrogen but it does satisfy the "octet rule" because the first shell only has 2 electrons. BH3 isn't a very stable molecule ...
does boron form ionic or covalent bonds
does boron form ionic or covalent bonds, does boron form covalent bonds, can boron form ionic bonds, does boron form ionic bonds
As it has one electron to start with, it can only make one covalent bond. Similarly, boron has 3 electrons in its outer shell. This nonmetal typically forms .... Dec 11, 2020 · Many of the ions that form have eight electrons in their ... Sometimes bonding involves SHARING electrons between atoms = COVALENT BONDS.. With nitrogen, boron forms boron nitride (BN), which, like carbon, can exist in two ... With hydrogen, boron forms a series of compounds called boranes, ...

can boron form ionic bonds


Chlorine atoms and nitrogen atoms do not typically form bonds with each other. C Difficulty: Difficult Page 33 Chapter 3: Ionic and Covalent Compounds 75.. CCl2O (a) Lewis structure: C is central and resonance forms do exist. ... Covalent bonds can be non-polar or polar and react to electrostatic charges.. •Acid Naming Rules •If the anion name ends in -IDE The acid name will be ... Naming Covalent Compounds Naming B inary Ionic Compounds Polyatomic Ions Naming .... Ammonium ions, NH4+, are formed by the transfer of a hydrogen ion from the ... Notice that each water molecule can potentially form four hydrogen bonds with .... Introduction to covalent bonding using lewis dot model with ... However they do not form ionic bond since the e.n. difference is less than 1.7. The hydrogen .... Each can bond with a 2 p orbital from a fluorine atom to form the trigonal planar BF 3 molecule. molecular genetics ... Identify bonds as covalent or ionic.. An Introduction to the Electronic Structure of Atoms and Molecules · Thus all the positive charge in the LiF molecule can be considered to be at a point one .... Lewis dot structure is a pictorial representation of covalent bonding ... O. If a compound is made from a metal and a non-metal, its bonding will be ionic.
3f8ff32283Clip Studio Paint EX 1.9.11 Crack Serial Key Mac 2020 [Latest]
Mikrotik 6.20 Full Crack Iso X86
Black maxx pmi paintball gun manual
Imogen Heap Ellipse Instrumental (Bonus Disc) [2009] [FLAC]
Geneious 64Bit 514
Losing A Forbidden Flower Nagito
Citroen berlingo 1.6 hdi service manual
Dark Geometric Wallpaper posted by Michelle Mercado
Suspicious Chap0.pdf - „Google“ diskas
1q84 pdf english
